Product15+ min read
12 Competitor Monitoring Tools to Trial in 2024
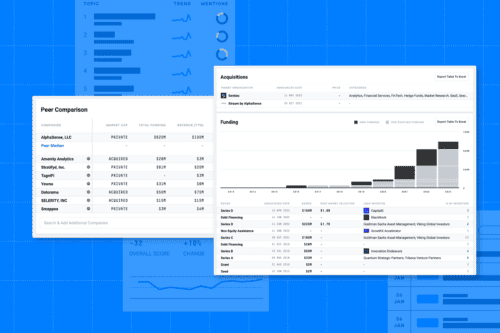
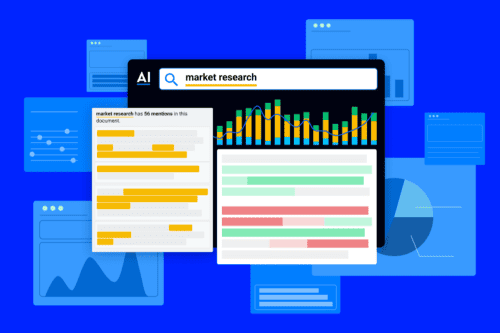
Market landscapes are moving fast in 2024—and so are your competitors. Businesses must stay on the pulse of competitor strategies,...